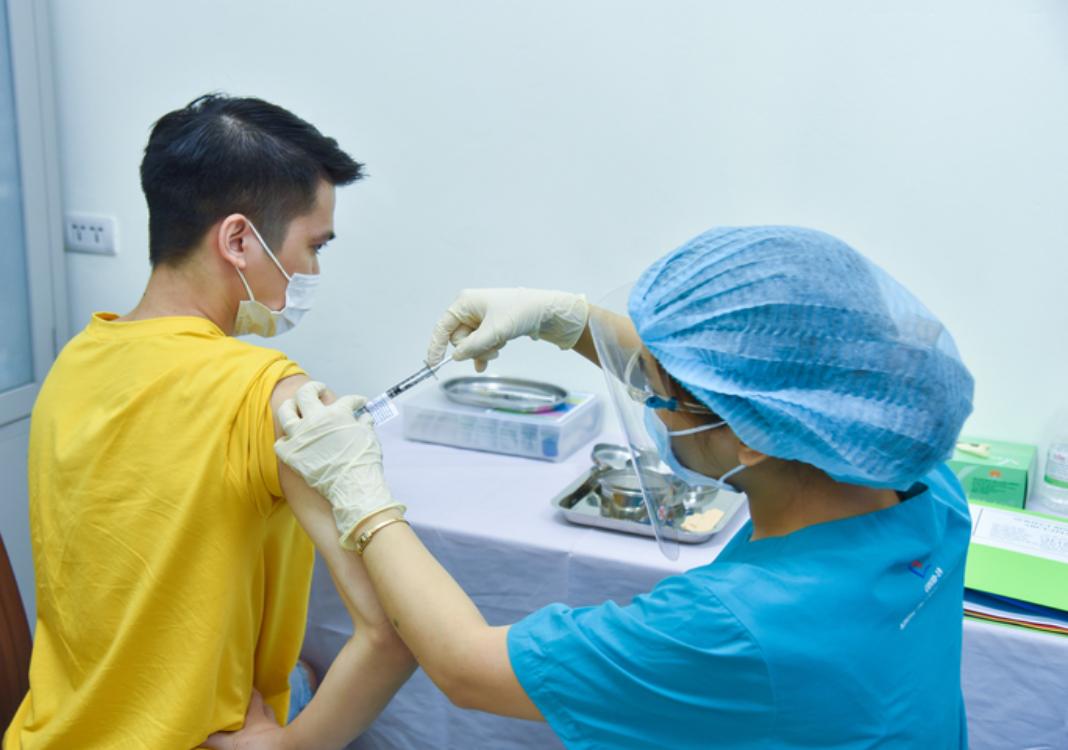
HCMC – Volunteers have been invited to sign up for phases 2 and 3a of clinical trials of the ARCT-154 Covid-19 vaccine candidate using the mRNA technology at the Hanoi Medical University.
From now until September 20, the university will receive registrations for participation in the clinical trials, the local media reported.
Volunteers should be 18 years old and above, reside in Hanoi City, should not have a medical history of viral respiratory diseases such as SARS, MERS and Covid-19 and should not have taken the Covid-19 vaccine.
Some 100 volunteers participating in the first phase of clinical trials of the home-grown vaccine candidate got a first shot at the research site of the university. The second shot will be administered to the 100 volunteers on September 12 and 13.
The result of the first phase of clinical trials showed that the ARCT-154 vaccine was safe.
ARCT-154 uses the mRNA technology and can self-multiply, so its immunity is expected to last longer. This is Vietnam’s first Covid vaccine candidate to apply the technology, which is used for the Pfizer and Moderna vaccines.
VinBioCare, a subsidiary of Vingroup, had earlier cut a deal with the U.S.-based Arcturus for the purchase of the technology to produce ARCT-154.
ARCT-154 is the third locally-made Covid vaccine candidate to begin human trials. All three phases of clinical trials of the vaccine will be executed in Vietnam.
Apart from the first phase with the participation of 100 volunteers, the second phase will be conducted with over 300 volunteers, while 20,600 volunteers will participate in the third phase of the clinical trial, with phase 3a needing 600 volunteers.
Earlier on August 2, the Ministry of Health had approved a plan to begin clinical trials of ARCT-154.